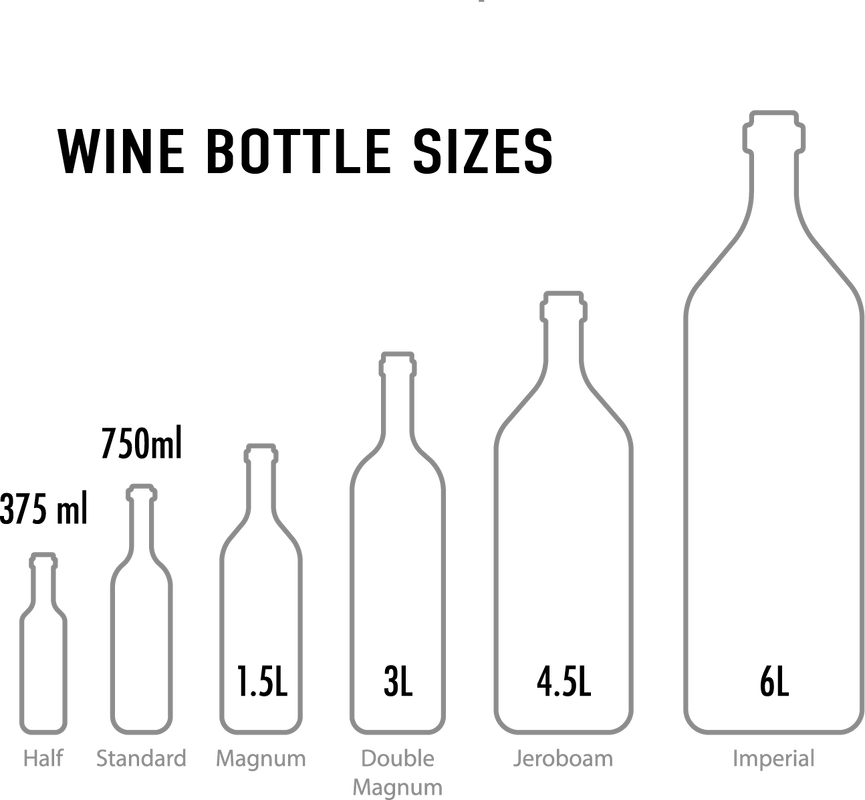
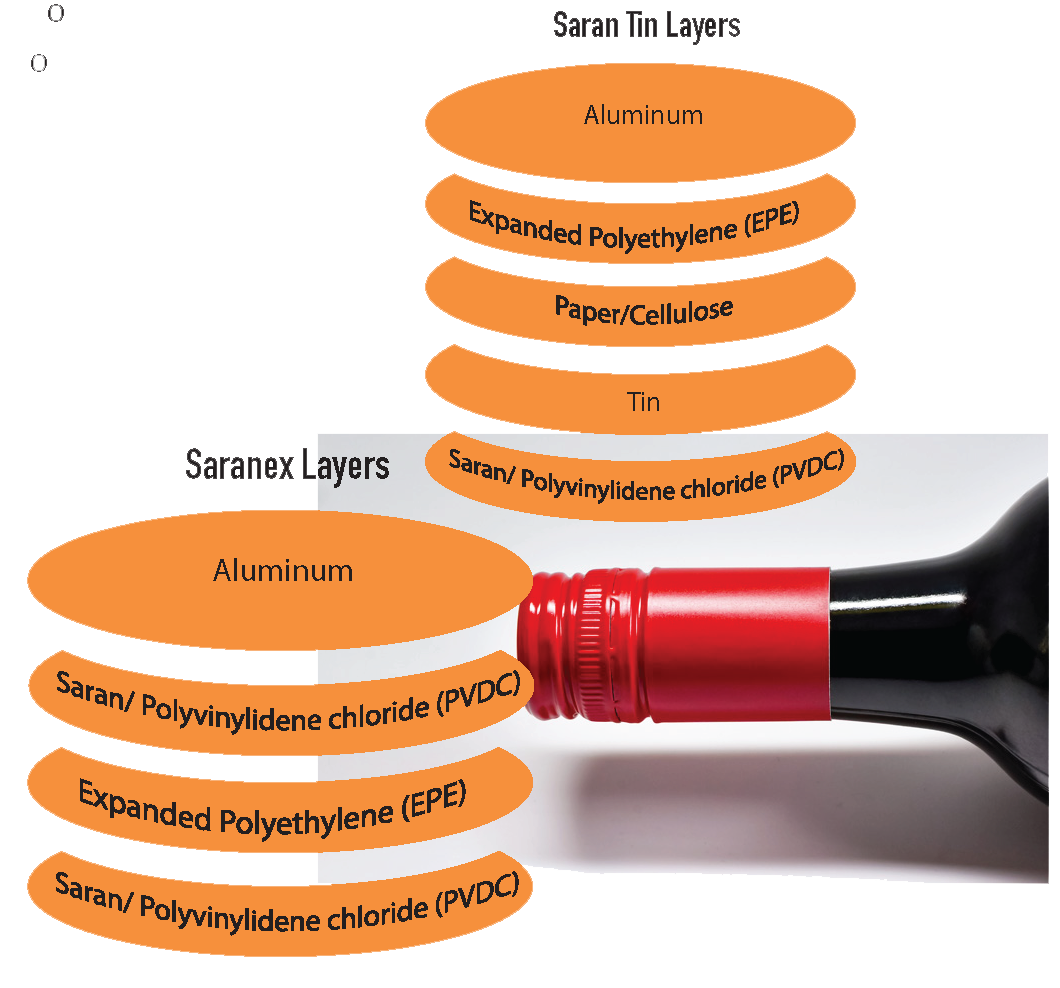
A Guide to: Wine Bottling
By: Brent Nakano
When bottling wine, the factors to take into account are making sure nothing gets out, that only the desired amount of oxygen gets in, and microbial stability. Then, during transport and storage, conditions should be adequate to support proper preservation and, if desired, proper development of the wine.